You are here
Iron and cancer, a balancing act

Cancer, the scourge of our century, continues to resist the therapeutic arsenal developed over the years by scientists throughout the world. The problem is that cancer is cunning. At present, most therapies preferentially target proliferating cancer cells. As well as enabling a tumour to grow, their rapid division induce selection pressure on their own community. This is why, within a tumour, some cells adapt and stop dividing, entering into a sort of dormancy that allows them to escape treatment.
Furthermore, this non-proliferative state is associated in some cases with other properties, such as an ability to migrate and invade other tissues – in other words, to metastasise. In addition, these metastatic cells are more resistant to current chemotherapies and have a high potential for colonisation. They are the subject of scientific studies by the CNRS, Institut Curie and INSERM, under the supervision of the chemist Raphaël Rodriguez1. “The main focus of our laboratory is to understand the nature of this adaptation and its underlying chemical and molecular bases,” he explains. “If we can elucidate how these cells adapt, we may be able to identify new targets and devise new drugs that will aim at and prevent these adaptations.”

The results of this approach, as novel as it is unique, have been published in the journal Nature2. It uses both a small molecule developed by the laboratory and a recently-demonstrated cell death process mediated by iron, called ferroptosis.
Iron is essential to life
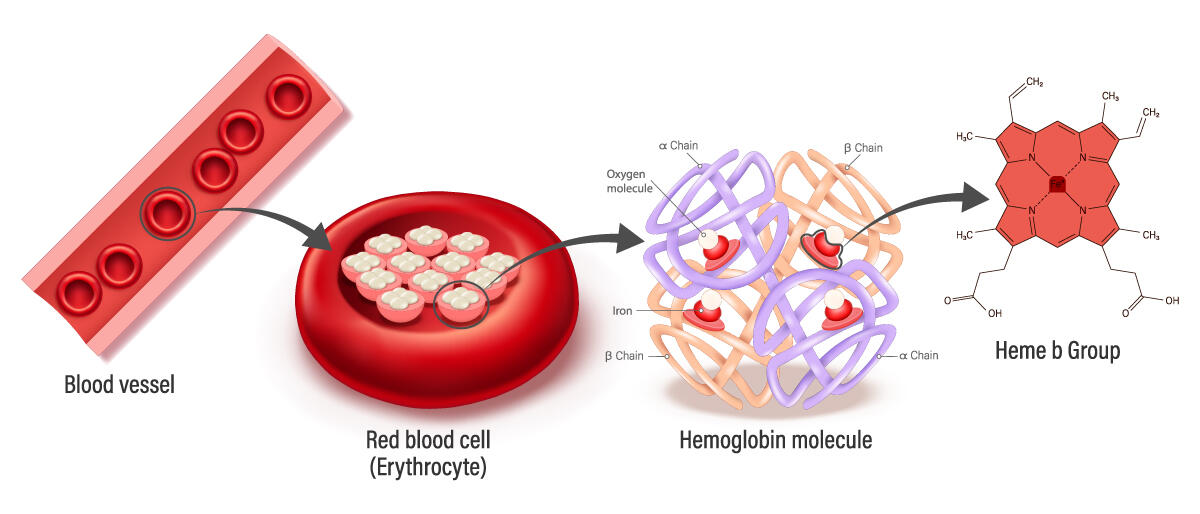
A metal present in vast quantities in the Earth’s crust, iron also plays an essential role in the living world – for example as an oxygen carrier via haemoglobin (giving it its characteristic red colour). More recently, it has been identified as a major factor in a type of cell death called ferroptosis.
“Ferroptosis refers to the vulnerability of cells to iron chemistry,” the scientist says. “In reaction to a high internal concentration of iron, the cell invents mechanisms to neutralise its harmful effects. If these mechanisms are still effective in a cancer cell, it will live, adapt and may metastasise. However, if the cell is not able to withstand the adverse effects of a high iron concentration (in this case, the production of oxygen free radicals), then it will die.”
More specifically, once iron has been internalised in a cell within a cellular compartment, it will react with the hydrogen peroxide that is present and produce oxygen free radicals. It is the latter that cause degradation of the lipids that make up the compartment membrane. This degradation then spreads gradually to all cell compartments (nucleus, mitochondria, etc.), which ultimately results in death of the cell through ferroptosis.
In other words, iron has a dual role here: it is essential to the transformation of primary tumour cells into metastatic cells, but in excessive quantities it induces their self-destruction by ferroptosis.
A new pathway for the internalisation of metals
“In 2020, we discovered a new pathway for the internalisation of metals into cells via the intervention of a membrane receptor called CD44,” Rodriguez explains. “We demonstrated that this receptor enables absorption of metals present in the immediate environment of a cell, and particularly iron and copper. And it is known that transforming cancer cells overexpress CD44.”
But what role does iron play in this mechanism? “To become metastatic, a primary cancer cell needs to change the expression profile of its genes. The cells in our bodies do not all express the same genes. Amongst other factors, their expression is regulated by the accessibility of certain genes to the transcription factors that define the identity and nature of a tissue.”

Iron enables catalysis of the accessibility of certain genes – and hence their expression –especially of those involved in the acquisition of resistance to standard treatments. Cancer cells therefore need iron to become metastatic. It is this phenomenon that the CNRS scientist is seeking to exploit in order to get rid of metastatic cells.
The mediator of ferroptosis
Indeed, during their transformation, cancer cells find themselves on a knife edge; to escape death by ferroptosis, they need to develop mechanisms that limit their intake of iron.
“Our aim is clear: it is to tip the scales towards their death rather than their dissemination. To achieve this, there is a narrow window to exploit this abundance of iron,” the chemist enthuses. “Our idea is to saturate the cell’s protective systems by exacerbating its reactivity to iron, in order to initiate the spread of changes to lipids.”
Based on their previous discoveries regarding the involvement of iron in ferroptosis, and their identification of the cell compartment in which intracellular iron is found, the scientists have developed a bifunctional molecule called fentomycin-1 (or Fento-1), “inspired by protein degraders such as the proteasome”.3



This new molecule is specific in that it possesses two different substrates. “Part of Fento-1 displays strong affinity for lipid membranes, and notably the most important of them, which forms the external envelope of all animal cells. It is known that this membrane will be internalised sooner or later, because its turnover is around two hours. In other words, the entire membrane is completely renewed every two hours,” the researcher reveals. “The second part of the molecule is a ligand 4 with strong affinity for iron, and a geometry and electronic density that favours iron chemistry. The whole molecule is able to induce the degradation of membrane lipids through the production of oxygen free radicals, and ultimately to trigger ferroptosis.”
Promising against sarcomas and pancreatic cancers
As effective in mouse models of metastatic cancer as in the primary cells of patients, organoids or biopsies of human tumours obtained just after surgery, Fento-1 also appears to be highly promising in the treatment of sarcomas and pancreatic cancers. In addition, according to Rodriguez, these findings are the result of a paradigm shift.
“This change is not based on using an anticancer agent or molecule with a particular mechanism, but on how we have now understood that it is the state of a cell that gives it specific vulnerability, and thus defines the type of molecule that can be used to fight it.”
In the longer term, the strategy could be to rely on a combination of anti-metastatic and anti-proliferative molecules to achieve optimal therapeutic responses. “All this is clearly based on new vulnerabilities of cancer cells,” concludes Rodriguez. “This has enabled us to improve our understanding of the biology of cancers, which deserves to be studied in greater depth before potential clinical applications.” ♦
See also
A new antibody for fighting cancer
Focusing on targeted treatments against cancer
- 1. CNRS research professor in the Chemical Biology of Cancer (CBC) laboratory (CNRS / INSERM / Institut Curie).
- 2. https://www.nature.com/articles/s41586-025-08974-4.
- 3. A multiprotein enzymatic complex present in the cytoplasm and involved in the specific degradation of poorly folded, denatured or obsolete proteins.
- 4. Molecule that can bind reversibly to a target (protein, amino acid, etc.).