You are here
Medical implants of the future

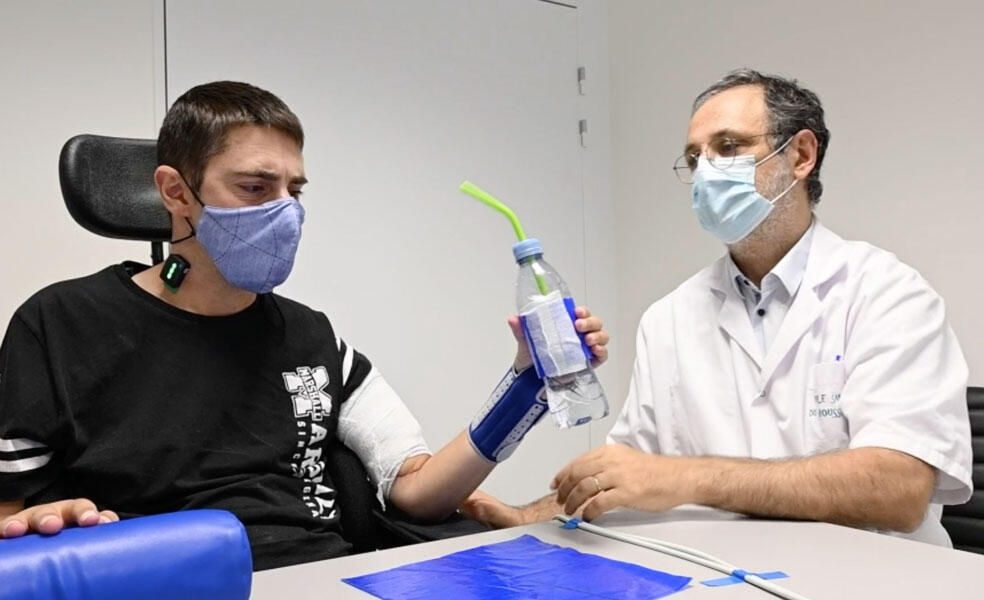
Imagine a quadriplegic patient holding their coffee cup, or a Parkinson’s sufferer toning down their tremors. It may seem like science fiction, but such achievements are made possible by the latest scientific advances in medical implants.
The functioning of these implants relies on bioelectronics. In the past ten years, great progress has been made in this field, whose goal is “to connect electrical systems with the human body for therapeutical or research purposes”, explains Denys Nikolayev1, a CNRS researcher at the IETR institute of electronics and digital technologies and author of a study2 on improving the power supply of implants.
Computer connection between the cortex and the spinal cord
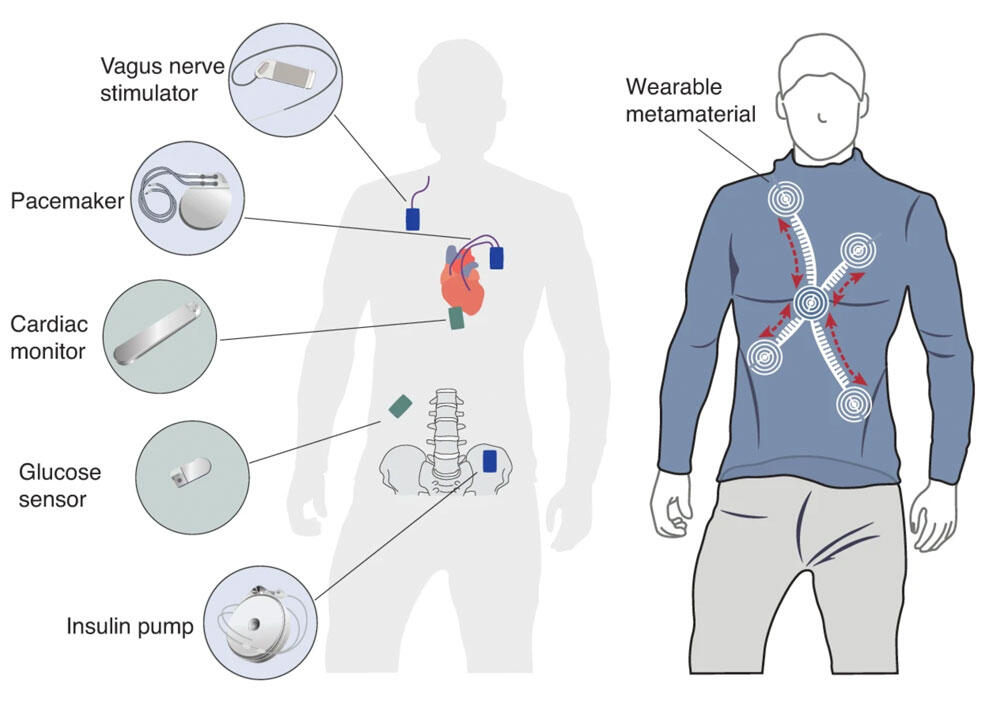
Since the invention of the pacemaker in the 1950s, a multitude of implants have been developed for an ever-wider range of applications. In 2023, a paraplegic patient was able to walk3 again thanks to the Wimagine4 implant, developed by the Clinatec research centre at the French Alternative Energies and Atomic Energy Commission (CEA). The device allows the brain to control a spinal cord stimulator developed by Swiss researchers at the EPFL Swiss federal technology institute and CHUV University Hospital in Lausanne.
Guillaume Charvet, head of the biomedical research department in neurotechnologies at Clinatec, explains how it works: “An implant on the surface of the brain records the electrical activity of the cortex, which is decoded by a computer, which in turn transmits commands to a stimulator positioned on the spinal cord, allowing the patient to move their legs.”

With the help of this device, patients are able to walk on their own, but remain dependent on a computer integrated in a walker. So far seven individuals have been equipped with the Wimagine implant in clinical trials.
Implants can also be used in other parts of the body. For example, the Neurinnov company enables quadriplegics to control their hands thanks to a device implanted under the skin of the chest, connected to electrodes that stimulate two nerves inside the arm. “Patients who had lost control of their hands can once again carry out simple gestures like picking up a cup or holding a bank card,” reports Neurinnov Chief Technical Officer David Andreu.
Induction powering
However, in order to work, these technological gems need power. Manufacturers have several options to choose from depending on the context. As Andreu explains, “Pacemakers perform a vital function. As a result, they cannot depend on an external energy source, and are therefore powered by a battery that has to be changed every eight to ten years.”
The use of a battery is a possible solution for non-vital implants as well, but it increases their volume. This implies more extensive surgery, putting additional strain on the patient. The battery also needs to be changed after a number of charge-discharge cycles (typically 300 to 500 for cochlear implants)5.

Some manufacturers offer battery-free implants powered continuously by an external antenna on the surface of the skin. These antennas work by induction: an electric current flows through a coil and creates a magnetic field, inducing a second current in the implant inside the body. The same principle is used for wireless phone charging, but the devices are somewhat different.
Still, the antenna that transmits the energy must itself be operated by a battery, with which it can be installed in a casing attached to the surface of the body. If more energy is needed – as is the case with the Clinatec and Neurinnov implants – the antenna is connected by wires to a larger battery, carried in a pocket, for example.
A theoretical framework as a manufacturing aid
Furthermore, as Nikolayev points out, powering implants by induction “is only feasible when these are large enough (about 2 to 5 cm) and relatively close to the body’s surface (ideally, within 2 cm)”.
The researcher is one of the authors of a study proposing a theoretical framework for optimising this power source. Their theory is based on electromagnetic waves, eliminating the need for wires and batteries. This is no simple task: there are significant losses due to the succession of tissues involved, each of which has different wave propagation properties.


The higher the relative permittivity of a medium, the shorter the wavelength and the slower the wave. For example, for a wave at 2.45 GHz, air has a relative permittivity of 1, while that of fat is 5 to 6, compared with 48 to 50 for the brain and 52 to 55 for muscle, Nikolayev reports. Consequently, “Compromises must be struck between the size of the implant, its depth, the frequency of the wave and the type of source to minimise these losses.”
The researcher and his colleagues offer equations for estimating each type of loss in relation to the characteristics of the implant. According to Nikolayev, “These equations can help implant manufacturers determine the optimal frequency and type of antenna for their devices, bypassing the need for complex, energy-consuming tests.”
In the study, the formulas were applied to the practical case of an ingestible capsule. The result was that the optimal frequencies yielded by the equations matched those determined by experimental tests on “body phantoms” reproducing human tissues.
Clinical constraints and standards
In addition to these technical restrictions, many other factors should be considered. As Charvet points out, “The device must take into account the medical need, the constraints of clinical operability and safety, as well as regulatory requirements such as long-term biocompatibility.”
The Wimagine implant has been developed to be minimally invasive and to enable chronic long-term use. “The device is made of titanium, placed on the surface of the brain without penetrating it,” Charvet explains. “The implantation surgery takes only an hour and it can be explanted.”
Since they receive and emit waves, these medical devices are subject to safety standards, just like telephones and microwave ovens. Some of these standards seek to limit heating of the tissues around the implant. Depending on which part of the body is concerned, the regulations define a specific absorption rate (SAR) to be met, corresponding to the maximum energy that the body can absorb. The SAR is then used to calculate the device’s ultimate power level.
The implants of tomorrow
In the years to come, Nikolayev is confident that medical implants will become “smaller, more flexible and biocompatible, which will allow them to function inside the body for decades”. Improved radiation supply of these devices, aided by theoretical studies, facilitates their miniaturisation and opens the way towards myriad possibilities.
In the longer term, groups of implants could communicate with one another. Nikolayev has participated in a study6 showing that a metamaterial textile (consisting of repeated conductive patterns) improved communication between a vagus nerve stimulator and a heart rate sensor by guiding waves across the body’s surface. A direct wireless network using this type of fabric to connect implants at the scale of the body has already been tested and validated in pigs.
Other research is looking even further ahead: scientists are studying wave transmission through adipose tissue to create networks of implants within the body7. Science fiction is coming true!
For further reading
- 1. IETR (CNRS / CentraleSupélec / INSA Rennes / Nantes Université / Université de Rennes).
- 2. G. Mingxiang, et al., “Physical insights and design principles for efficient wireless implantable bioelectronics,” Cell Reports Physical Science, 2025: https://tinyurl.com/implants-bioelec
- 3. H. Lorach, et al., “Walking naturally after spinal cord injury using a brain–spine interface,” Nature, 2023: https://doi.org/10.1038/s41586-023-06094-5
- 4. C. S. Mestais, et al., “WIMAGINE: wireless 64-channel ECoG recording implant for long term clinical applications,” IEEE Transactions on Neural Systems and Rehabilitation Engineering, 2014: https://doi.org/10.1109/TNSRE.2014.2333541
- 5. The number of cycles varies according to the brand of implant, temperature, frequency of discharges, discharge rate at the start of charging, etc.
- 6. X. Tian, Q. Zeng, S. A. Kurt, et al., “Implant-to-implant wireless networking with metamaterial textiles,” Nature Communications, 2023: https://doi.org/10.1038/s41467-023-39850-2
- 7. N. B. Asan, et al., “Characterization of the Fat Channel for Intra-Body Communication at R-Band Frequencies,” Sensors, 2018: https://doi.org/10.3390/s18092752